|
4/12/2023 0 Comments Define ion bonding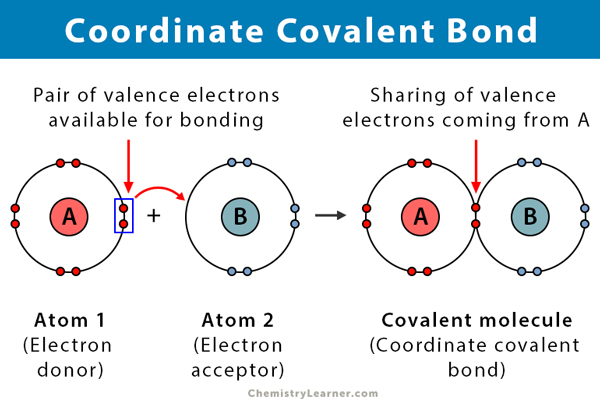
Ionic bonds share the electrons very unequally and can be broken into ions. Covalent bonds share the electrons more equally and are stronger. An ionic bond forms when two ions of opposite charges exchange electrons between them, where an ion is an atom that has either lost or gained an electron. When the electrons being shared are so unevenly distributed between the atoms the bond that is formed is called an ionic bond. Ionic and covalent bonds differ in the degree of sharing of the electron densities between them. Atoms involved in covalent bonding do not separate into ions. This creates a stronger bond between the two atoms that is more difficult to separate. Ionic compounds are pure substances formed from chemically bonded ions. 
The electron density is more evenly shared between the two atoms. In covalent bonds there is no difference or a small differences in electronegativity. When such an electron transfer occurs, one of the atoms acquires a negative charge and is dubbed the anion. The ionic bond is defined as the electrostatic forces that bind together oppositely. In this case, an atom loses an electron, which is then gained by another atom. Electropositive and electronegative chemical elements form an ionic bond due to the transfer of electrons from electropositive to electronegative atoms forming ions that are bound by strong electrostatic forces to form ionic compounds. This makes it easy for the two atoms involved int the bond to separate leaving one atom with a positive charge and one atom with a negative charge. Ionic bonding is a type of chemical bonding in which electrons are transferred from one atom or molecule to another. In Ionic bonds there is a large difference in electronegativity between the two atoms ( greater than 1.7) The large difference in electronegativity results in most of electron density being located next to the atom with the greatest electronegativity, and very little electron density being next to the atom with the lower electronegativity. This bonding refers to the complete transfer of valence electrons. An ionic bond can form between two elements when one has a small number of electrons in the valence shell (metal) and one has an almost full outer shell. Ionic bonds can occur between metals and. The electron density of the atoms is held between the two atoms and attracted to the positive forces of both sets of protons in the two nuclei. Ionic bond refers to a type of chemical bond which generates two oppositely charged ions. An ionic bond is the electrostatic force of attraction between two oppositely charged ions (called anions and cations). 
Chemical bonds are formed by the sharing of electron densities between two atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
